(GW Pharmaceuticals)
The medical marijuana drug Sativex, which could be approved in the United States in the coming years as a treatment for pain relief, has little potential for abuse, experts say.
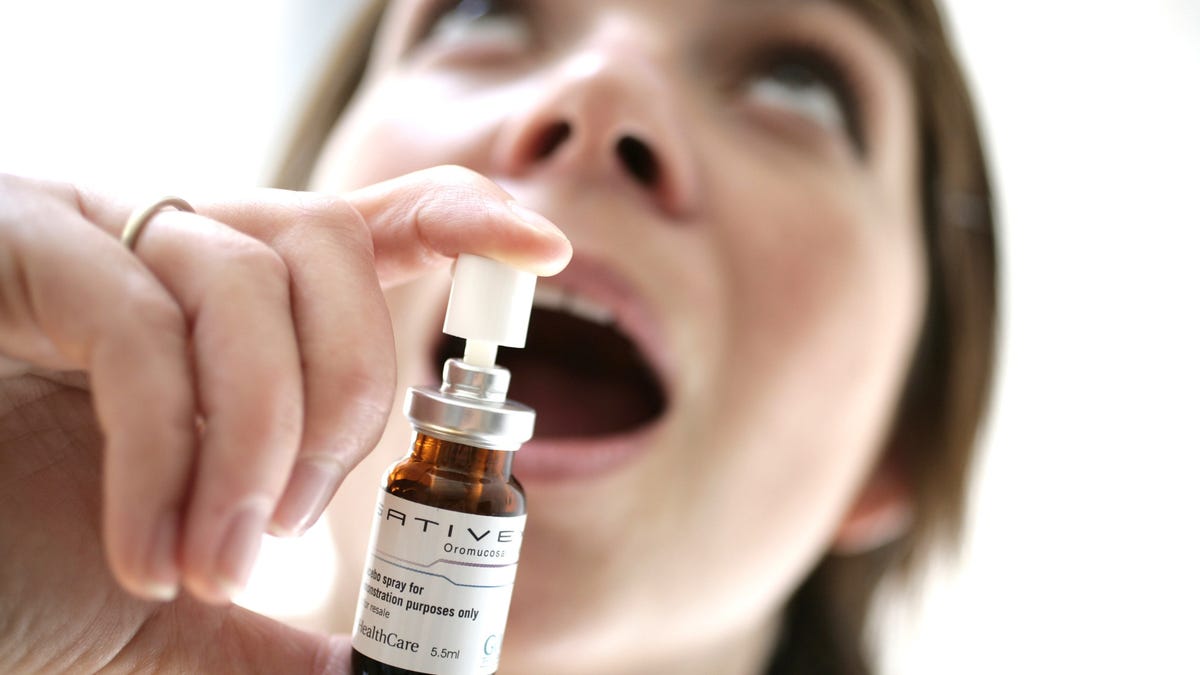
The British pharmaceutical company GW Pharmaceuticals is currently testing the drug, which is delivered as a mouth spray and called Sativex, in clinical trials. The company plans to seek U.S. Food and Drug Administration approval for the drug as a treatment for cancer pain when the trials are completed, likely sometime in 2014, a spokesperson for GW Pharmaceuticals told MyHealthNewsDaily.
The active ingredients in Sativex, known as cannabinoids, are derived from the cannabis plant. It is the first marijuana-based drug to be made by extracting the compounds from the plant, rather than synthesizing them. Two other drugs, Marinol and Cesamet, based on synthetic cannabinoids, were approved by the FDA in the 1980s.
Because the drug contains THC, the ingredient primarily responsible for marijuana's "high," it's possible people would use the drug for recreational rather than medical purposes.
"There is no doubt in my mind that there will be people that abuse it," said Dr. Jeffrey Bernstein, director of the Florida Poison Information Center at the University of Miami Miller School of Medicine.
However, because the drug is delivered through ingestion, rather than smoking, it would take much longer to have an effect — at least an hour, compared with the minutes it takes to get high after smoking marijuana, said Margaret Haney, a professor of clinical neurobiology at Columbia University. This means drug users seeking a high would be less likely to abuse it. "Smoking is a really effective way to get a chemical into the brain," Haney said. The mouth spray "is a far safer administration,"she said.
And Marinol and Cesamet, which are also administered orally, have a low rate of abuse. "We don’t see a lot of problems from [those]," Bernstein said.
Not the same high
GW Pharmaceuticals intends to market Sativex in the United States for treatment of cancer pain. The drug is already approved in United Kingdom, Spain, Canada and New Zealand to treat muscle spasms due to multiple sclerosis, according to the company website.
Patients can adjust the dose of Sativex to prevent it from entering the blood too rapidly, allowing them to experience symptom relief without the marijuana high, according to GW Pharmaceuticals.
In addition, while marijuana is a hodgepodge of about 64 different substances, Sativex is composed mainly of two ingredients: THC and another cannabinoid called CBD. The latter component is thought to ameliorate some of the side effects of THC, including the high that marijuana users feel, said Dr. Armando Villarreal, an assistant professor of neurosurgery and pain management at the University of Rochester Medical Center in New York.
And for habitual marijuana users, the cultural and ritualistic practices that go along with smoking pot, such as passing a joint, may be an important part of the experience, Bernstein said. These rituals cannot be replicated with the spray. "A lot of people that smoke marijuana would rather smoke it," he said.
Unlikely overdose
Unlike drugs such as painkillers, which come with a risk of death if people take too much, patients who "overdose" on the marijuana spray would be at little risk for acute health problems, Haney said.
"What could happen is the person could get very uncomfortably intoxicated," Haney said. But in terms of other serious health effects, "there's none that I know of," Haney said.
"Marijuana, in the scheme of things, is a relativity safe drug," Bernstein said. "Even as a smokeable drug of abuse, it's relatively safe…compared to cocaine or heroin."
However, Villarreal noted that for people with psychiatric disorders, smoking marijuana has been shown to make the patients' mental problems worse. It's possible Sativex may also cause this problem in some patients, he said.
Could it help patients?
So far, the studies that have been conducted do not provide enough evidence to say Sativex is effective in improving pain symptoms, Villarreal said.
Sativex has mainly been tested as a drug to treat pain caused by damaged nerves. If the drug is approved by the FDA, Villarreal speculated, its use could be restricted to certain pain patients. Those with cancer pain that is not caused by damaged nerves may not be candidates for the drug, Villarreal said.
About 1 in 11 people who try pot end up addicted to it, Haney said. It would be interesting to study whether Sativex could help people in dependent marijuana users quit the drug, she said.